
Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.
Select Language

English
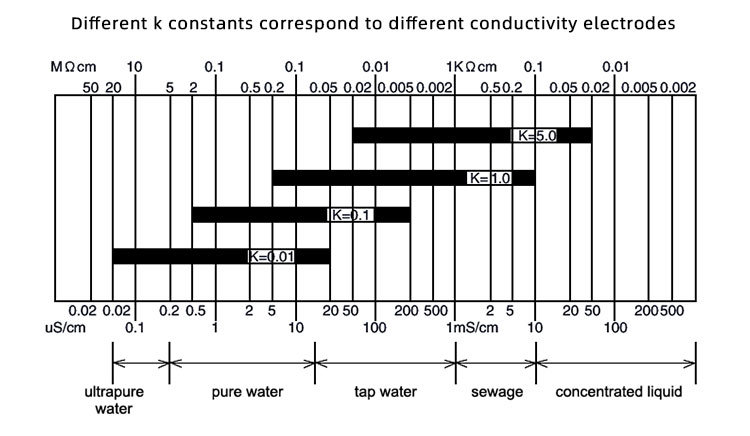
In the realm of electrochemical measurements, the K constant (or slope) of conductivity electrodes plays a pivotal role. It defines the linear relationship between the electrode's response and the sample's conductivity. Different K constants correspond to various conductivity electrodes, and selecting the appropriate one is crucial for obtaining accurate measurement results.
?
Conductivity (κ) measures a substance's ability to conduct electricity, typically in S/cm (Siemens per centimeter). Conductivity electrodes assess the ion concentration in a sample to determine its conductivity. The K constant reflects the electrode's sensitivity, with common values like 0.01, 0.1, and 1.0. A higher K constant indicates greater sensitivity to changes in the sample's conductivity.
From the graph, it's evident that electrodes with different K constants are suited for various conductivity ranges, ensuring more accurate measurements. For instance:
Conductivity electrodes are made from various materials, each with its own set of properties that make them suitable for different applications. One of the most common materials is stainless steel.
Stainless steel electrodes are known for their durability and corrosion resistance. They are often used in industrial settings where the environment may be harsh or the sample may contain corrosive substances. Stainless steel electrodes can withstand high temperatures and pressures, making them ideal for use in processes where these conditions are present.
In addition to stainless steel, other materials used for conductivity electrodes include platinum, titanium, and graphite. Each material has its own advantages and disadvantages, and the choice of material will depend on the specific application and the properties of the sample being measured.
?
Here are some typical application environments based on conductivity ranges:
Selecting the appropriate conductivity electrode is vital for measurement accuracy. Consider the sample's conductivity range, the environment in which the measurement will be taken, and the properties of the available electrode materials when choosing the K constant and electrode type. If the range is unknown or varies widely, electrodes with a moderate K constant, like K=0.1, and a durable material like stainless steel offer a good balance.
In conclusion, understanding the relationship between K constants, conductivity electrodes, and their materials is essential for accurate measurements across different conductivity ranges and application environments. By applying this knowledge, you can significantly enhance the accuracy and reliability of your electrochemical measurements.
Keywords: K constant, conductivity electrode, electrochemical measurements, electrode selection, application environments, electrode materials, stainless steel electrode
September 25, 2024
August 27, 2024
November 07, 2023
October 20, 2023
In the field of water quality monitoring and analysis, Daruifuno's water quality instrument development team has stringent requirements on the performance of sensors, especially for waterproof...
Overview As a high-precision water quality monitoring device, the signal detection process of fluorescent dissolved oxygen sensor involves knowledge and technology in multiple fields such as optics,...
Introduction to Electrode Glass Membrane The electrode glass membrane, a critical component in pH electrodes, possesses a series of unique properties that make it suitable for use in harsh...
Overview Water quality is a critical factor in environmental protection, industrial processes, and public health. Effective monitoring and control help maintain safe drinking water, optimize...
Email to this supplier
September 25, 2024
August 27, 2024
November 07, 2023
October 20, 2023

Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.

Fill in more information so that we can get in touch with you faster
Privacy statement: Your privacy is very important to Us. Our company promises not to disclose your personal information to any external company with out your explicit permission.