Overview of pH
pH generally refers to the hydrogen ion concentration index. Hydrogen ion concentration is a method of expressing hydrogen ion concentration. It is the negative value of the common logarithm of the hydrogen ion concentration (activity) in aqueous solution, that is, -lg[H?]. The determination of hydrogen ion activity index can be qualitatively determined by using pH indicators and pH test paper, while quantitative pH measurement requires the use of pH analyzers or pH meters.
?
Origin of the pH Concept
The hydrogen ion concentration index, generally referred to as "pH", was proposed by Danish biochemist Soren Peter Lauritz Sorensen in 1909. The symbol p comes from the German word Potenz, which means "concentration". H stands for hydrogen ion.
For information, please refer to Wikipedia - https://en.wikipedia.org/wiki/PH#History
?
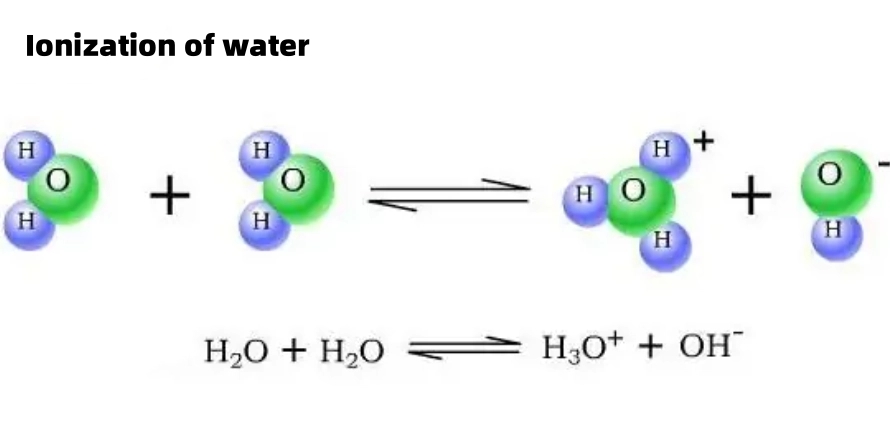
Ionization of Water and pH
As an extremely weak electrolyte, water can undergo weak ionization. Its equation is abbreviated as H?O?H?+OH?. This process is endothermic and affected by temperature. Acidic and alkaline environments will inhibit the ionization of water. At 25℃, only a small amount of water molecules in pure water are ionized, and the number of ionized H? and OH? is always equal, with a concentration of 1×10??mol/L. The ion product constant KW ([H?]×[OH?]) of water only changes with temperature and is a temperature constant. For example, KW=1×10??? at 25℃ and 1×10??? at 100℃. The acidity and alkalinity of a solution is determined by the relative concentrations of [H?] and [OH?], but it is inconvenient to compare directly at low concentrations. Therefore, the pH value is introduced and defined as the negative logarithm of the hydrogen ion concentration (mol/L), that is, pH=-lg[H?], which is convenient for measuring the acidity and alkalinity of a solution.
?
?
Methods for Determining pH
There are many methods for determining the pH of a solution:
1. Use a pH indicator. Add a pH indicator to the solution to be tested. Different indicators will change color according to different pH values. The pH range can be determined based on the study of the indicator. During titration, an accurate pH standard can be made.
2. Use pH test paper. There are wide-spectrum test paper and precision test paper. Use a glass rod to dip a little of the solution to be tested onto the test paper, and then compare the color change of the test paper with the standard colorimetric card to get the pH of the solution. pH test paper cannot show the pH of oil, because pH test paper measures the pH of the solution to be tested with hydrogen ions, but oil does not contain hydrogen ions, so pH test paper cannot show the pH of oil.
3. Use a pH analyzer or pH meter. Both pH analyzers and pH meters are instruments for measuring the pH of solutions. They measure the pH of solutions through pH selective electrodes or pH glass electrodes.
?
?
Importance and Applications of pH Measurement
Accurately express the pH of a solution
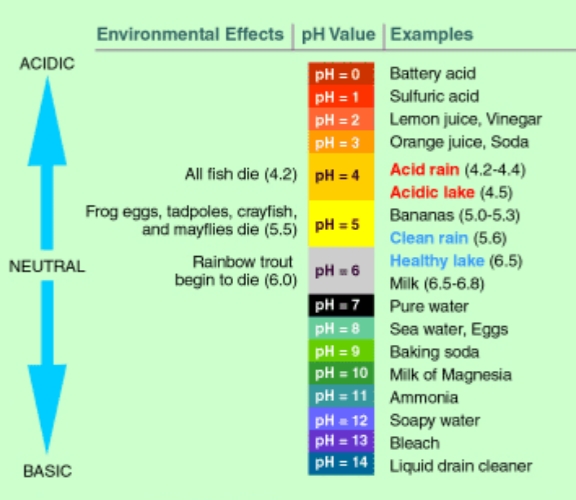
At standard temperature (25°C) and pressure, an aqueous solution with a pH of 7 (e.g., pure water) is neutral because the product of the concentrations of hydrogen ions and hydroxide ions naturally ionized by water at standard temperature and pressure (the ion product constant of water) is always 1×10???, and the concentrations of both ions are 1×10??mol/L. A small pH value indicates that the concentration of H? is greater than that of OH?, so the solution is highly acidic; a large pH value indicates that the concentration of H? is less than that of OH?, so the solution is highly alkaline. Therefore, the smaller the pH value, the stronger the acidity of the solution; the larger the pH value, the stronger the alkalinity of the solution.
At 25°C, when the pH value is less than 7, the solution is acidic; when the pH value is greater than 7, the solution is alkaline; and when the pH value is 7, the solution is neutral. Therefore, at room temperature, the pH value is usually a number between 0 and 14 (the pH value of concentrated sulfuric acid is about -2). However, in non-aqueous solutions or under non-standard temperature and pressure conditions, pH = 7 may not mean that the solution is neutral. This requires calculating the ionization constant of the solvent under such conditions to determine the neutral pH value.
?
?
pH is one of the most important physical and chemical parameters of aqueous solutions. All natural phenomena, chemical changes and production processes involving aqueous solutions are related to pH. Therefore, pH measurement is required in industry, agriculture, medicine, environmental protection and scientific research.
?
Daruifuno's Commitment to pH Measurement
As a professional water quality analysis instrument manufacturer, Daruifuno is committed to the research and development and production of high-quality pH analyzers, PH ORP analyzers, PH ORP controllers, pH sensors and other products. Our products are efficient, accurate and stable, and are widely used in industrial online, laboratory and portable occasions. If you have water quality monitoring needs or are interested in our products, we will serve you wholeheartedly!
?
?